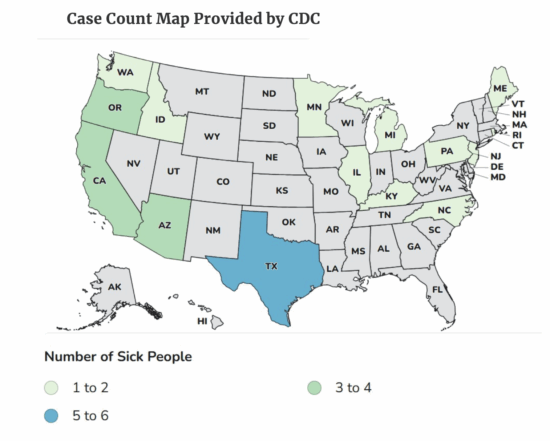
The patient count in an outbreak of infant botulism linked to ByHeart formula is continuing to increase, and federal officials say recalled product is still available in some stores.
As of Nov. 19, there are 31 sick babies from 15 states in the outbreak. All of them have required hospitalization. Illness onset for the patients has been from Aug. 9 through Nov. 13. The patients have been confirmed to have been fed ByHeart infant formula.
There has been an uptick of infant botulism cases since Aug. 1, with 84 patients having been identified. More of those patients will likely be identified as part of the ByHeart outbreak as testing continues.
ByHeart has recalled all of its infant formula products, but the Food and Drug Administration reports that some of the products are still available in some stores. The formula is sold in stores and through online marketplaces.
Public health officials are urging parents and care givers to not use ByHeart infant formula. Parents and care givers should watch for signs of infant botulism and immediately seek medical attention if any of the symptoms are observed.
Symptoms include constipation, poor feeding, loss of head control, and difficulty swallowing, which can progress to difficulty breathing and respiratory arrest. Symptoms of infant botulism, which is diagnosed clinically, can take as long as several weeks to develop following formula ingestion.
The formula was shipped to consumers worldwide. The U.S. FDA is warning parents and care givers around the world to not use ByHeart formula products.
As part of the outbreak investigation, public health officials in several states have collected leftover infant formula for testing. On Nov. 8, the California Department of Public Health (CDPH) preliminarily detected the presence of botulinum neurotoxin type A in a culture of ByHeart powdered infant formula from an opened can that was fed to an infant with laboratory confirmed infant botulism. Additional work by CDPH confirmed the identification of the bacterium, Clostridium botulinum type A, from the culture.
As part of its onsite inspections at ByHeart production plants, FDA collected and is analyzing samples of unopened product. Analysis is ongoing and results will be made public as they become available.
Additional testing is underway, and results are expected in the coming weeks. Detection of Clostridium botulinum in infant formula is difficult, and a negative test result does not rule out the presence of the bacteria in the product, according to the FDA.
The California Department of Public Health has opened a public hotline for concerned parents and caregivers with questions about infant botulism. The public can reach CDPH directly toll-free at 833-398-2022.
The public hotline is open seven days a week from 7 a.m. to 11 p.m. Pacific Standard Time (PST). Trained public health staff will be able to provide answers and information for concerned members of the public with questions about the current outbreak.
History of problems
The problems at one ByHeart production facility have been going on since at least 2022, according to an FDA warning letter sent to the company on Aug. 2, 2023.
In the warning letter, the FDA pointed out that the company had not established a system of process controls designed to ensure that infant formula in the production plant does not become contaminated with microorganisms. The FDA also noted that on Oct. 22, 2022, the company reported to (redacted) that a batch of ByHeart formula had tested positive for Cronobacter. Cronobacter can cause serious infections in babies that are often fatal.
At first, the positive test result showing contamination with Cronobacter was said to be a lab error, but further investigation showed this to not be the case.
“the third-party laboratory provided an ‘OOS Result Investigational Report for Microbiological Testing’ regarding the Cronobacter sakazakii findings in finished product, which concluded that ‘Laboratory error was not found or supported,’ “according to the warning letter.
“The third-party laboratory reached this conclusion after reviewing its internal protocols and testing conditions, finding no basis to attribute the sample results to laboratory error. Typically, a conclusion of lab error within an RCA is supported with confirmed evidence, such as written confirmation from the third-party laboratory attesting to the error. Here, there is no attestation; there is evidence to the contrary.
“Despite the discrepancy between the third-party laboratory and the internal conclusion within (redacted) root cause analysis, neither you nor your subsidiary company, (redacted), took any additional efforts to evaluate other routes of contamination that may have contributed to this event. Our review of your records obtained during the (redacted) inspection show that you did not work with your third-party contract manufacturer to further investigate the origin(s) or root cause(s) of the finished product positive findings. As the parent company and entity making all product disposition decisions, it is your responsibility to investigate all aspects of the production process for your products, including operations conducted at your (redacted) facility and your third-party contract manufacturer’s facility, when evaluating the potential root cause(s) for a product contamination event.”
(To sign up for a free subscription to Food Safety News, click here)